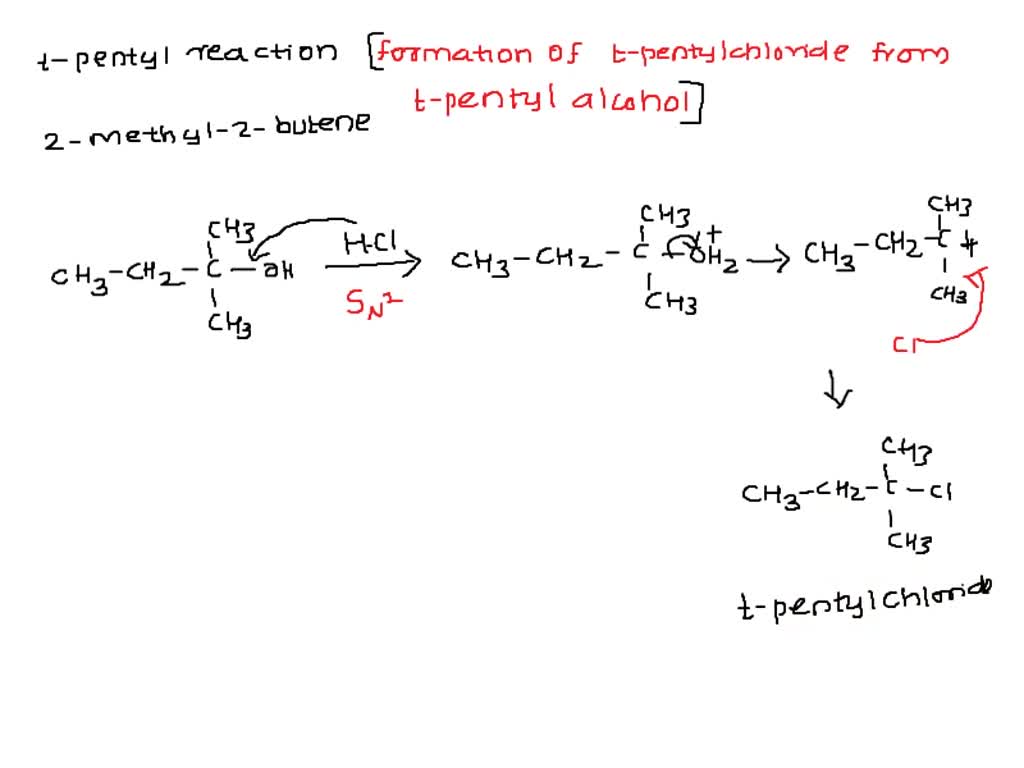
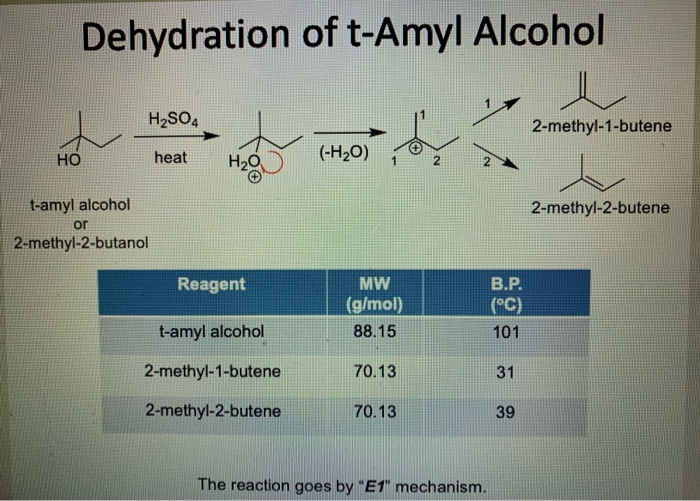
✓ Solved: t -Pentyl Chloride Some 2-methyl-2-butene may be produced in the reaction as a by-product....

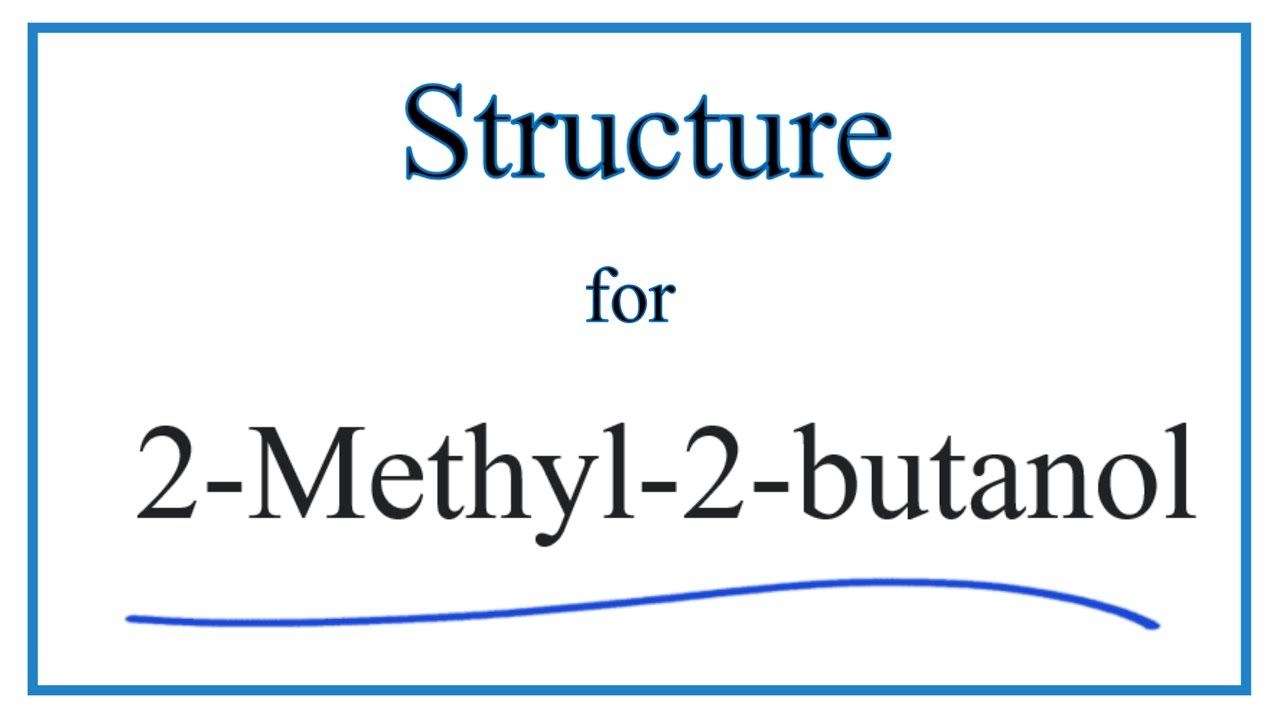
OneClass: Ifthe reaction of 2 methyl 2-butanol + HCl to give 2-chloro-2-methylbutane is conducted in ...
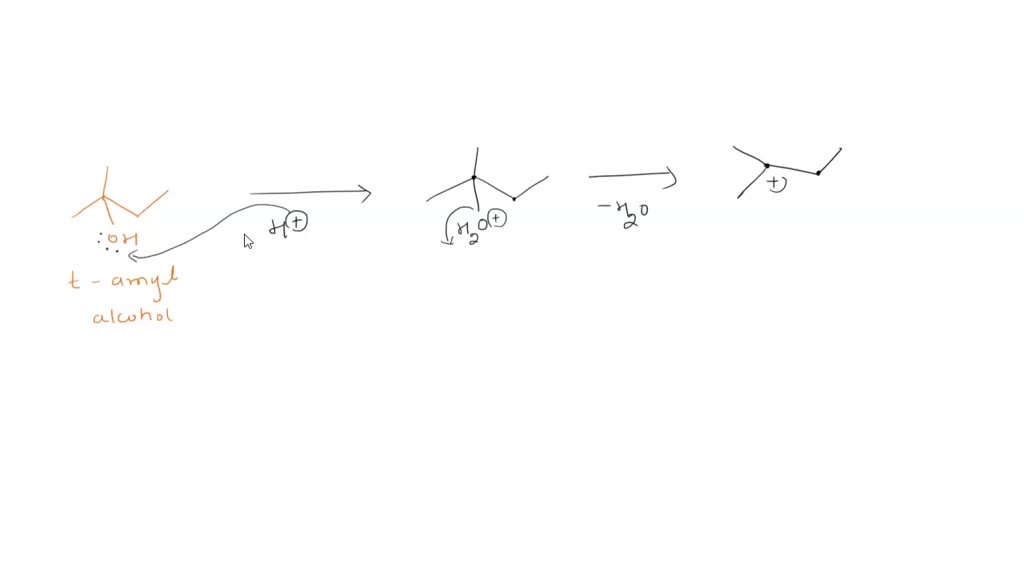
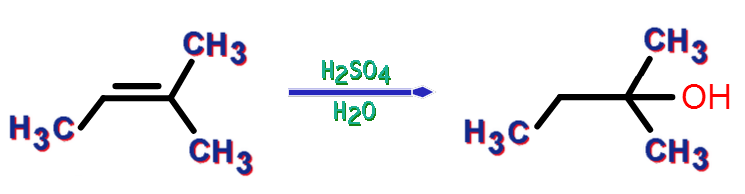
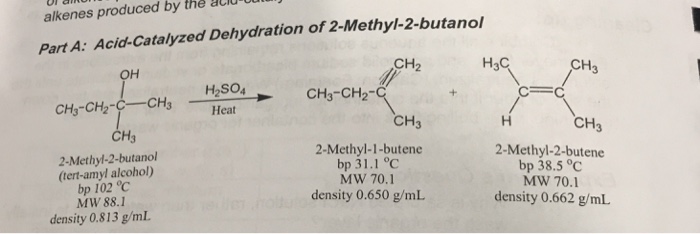
SOLVED: Draw mechanisms for formation of possible alkenes obtainable from t-amyl alcohol. Explain why 2-methyl-2-butene is the alkene that is obtained.

1) Show the mechanism for the conversion of 2-methyl-2-butanol into 2 -chloro-2-methyl butane. Remember to show the movement of electron pairs. 2) What type of reaction could occur if the alcohol and H

What is the major product obtained from hydroboration-oxidation of 2-methyl- 2-butene? | Homework.Study.com

Acid-catalyzed dehydration of 3-methyl-2-butanol gives three alkenes: 2- methyl-2- butene, 3-methyl-1-butene, and 2-methyl-1-butene. Propose a mechanism to account for the formation of each product. | Homework.Study.com
What would be the major product obtained from hydroboration–oxidation in 2- methyl-2-butene? | Socratic
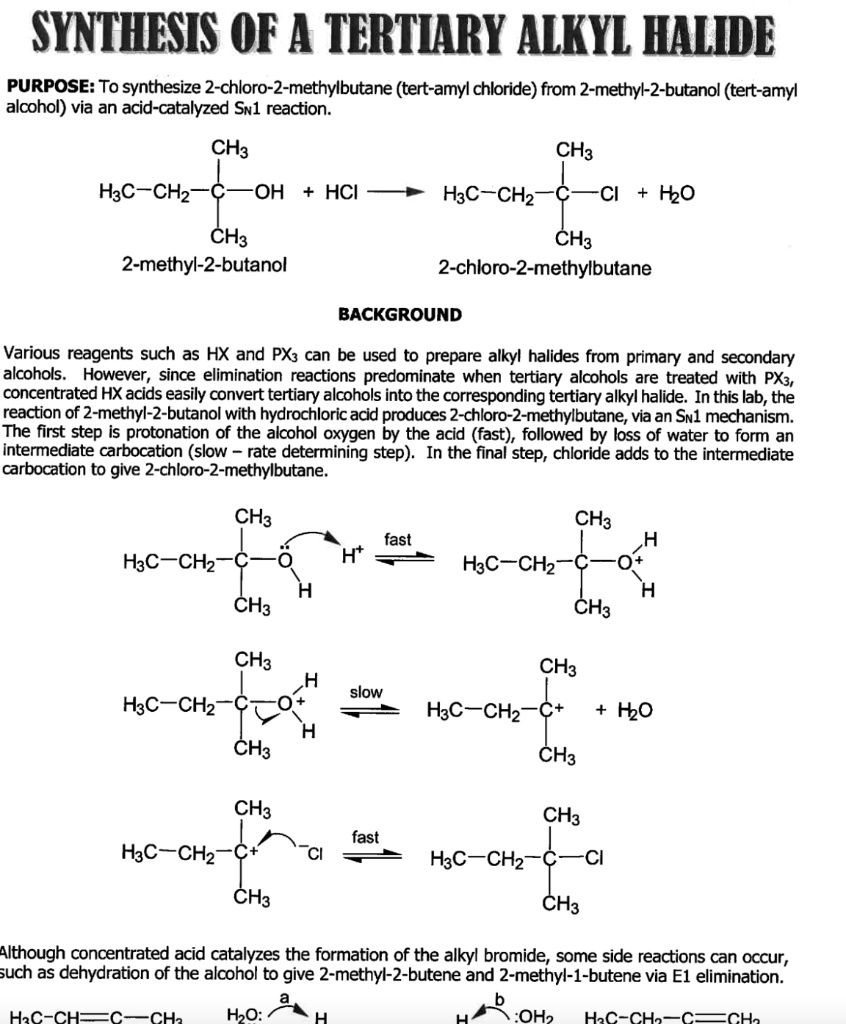
SOLVED: SYNTHESES OF A TERTIARY ALKYL HALIDE PURPOSE: To synthesize 2 -chloro-2-methylbutane (tert-amyl chloride) from 2-methyl-2-butanol (tert-amyl alcohol) via an acid-catalyzed Swl reaction: CH3 CH3 H3C-CHz OH HCI H3C-CHz Cl +2O CH3 CH3